AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Ammonium ion bonding1/30/2024 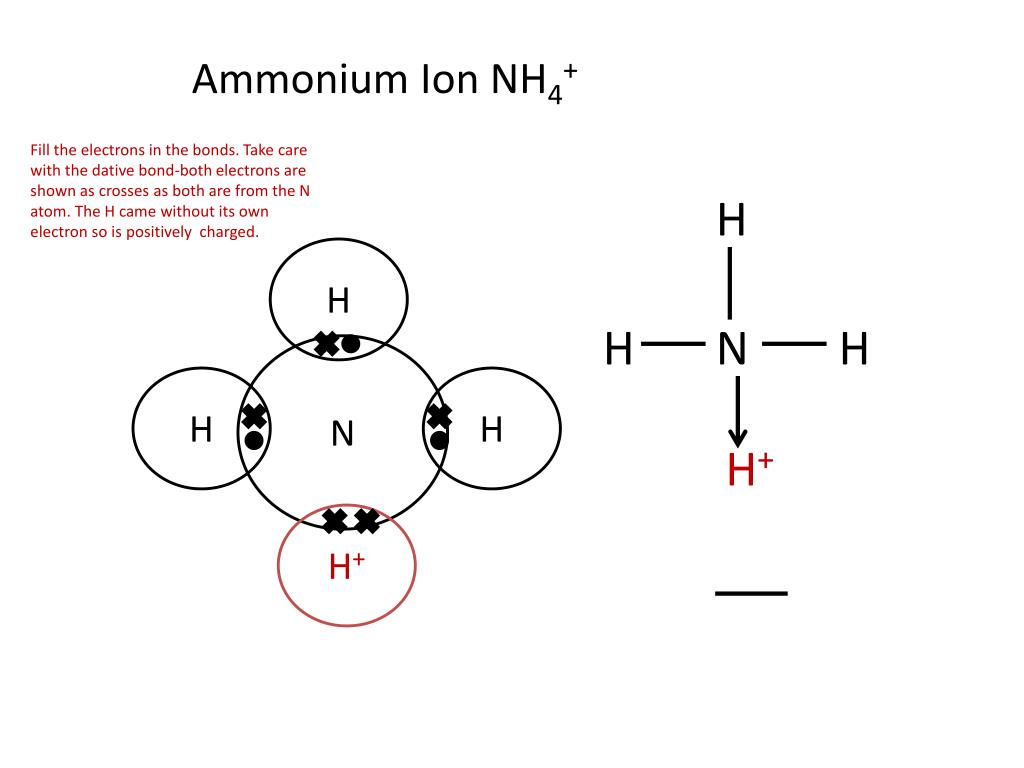
Moreover, complexes involving multiple hydrogen bonds may have slower dissociation kinetics, allowing competition from fast dissociation processes that have substantial energy barriers. In contrast, those complexes strongly bound by multiple hydrogen bonds dissociate not only to the protonated polyether and/or ammonium ions but also by extensive covalent bond cleavage of the protonated ether skeleton.This latter type of dissociation behavior suggests that the polyether/ammonium ion complexes may be sufficiently strongly bound that surpassing the high barrier to decomposition results in formation of internally excited polyether molecules that may then undergo subsequent fragmentation by skeletal cleavages. After collisional activation, those complexes that are weakly bound dissociate to form intact protonated polyether molecules and/or ammonium ions by simple cleavages of the hydrogen-bond association interactions. The nature of the hydrogen-bonding interactions of the ion complexes are evaluated by comparison of their collision-activated dissociation spectra. Ammonium ion (NH4+) The reaction of ammonia with an acid to produce ammonium salt fits in the examples of a coordinate covalent bond formation. In this study a variety of hydrogen-bonded crown ether/ammonium ion complexes are generated in the gas phase by association reactions between an amine substrate and a polyether, one of which is initially protonated, and stabilized by many collisions in the chemical ionlzation source of a triple quadrupole mass spectrometer or in a quadrupole ion trap. This study presents the first non-metal battery with potential for low-temperature applications and opens the door to future metal-free electronics that would generate long-term benefits to the environment.Hydrogen bonds are among the most important interactions involved in selective complexation in host-guest chemistry. Ammonium ions, NH 4+, are formed by the transfer of a hydrogen ion from the hydrogen chloride to the lone pair of electrons on the ammonia molecule. 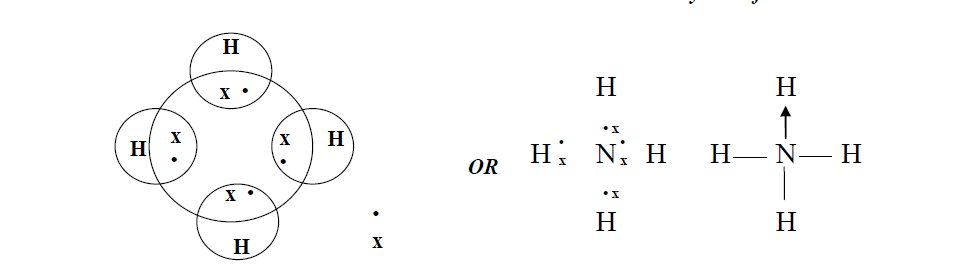
Additionally, the physiochemical properties of NH 4 +-based WiSEs are examined by Raman and nuclear magnetic resonance (NMR) spectroscopies, to explore their electrochemical behaviors and the fundamental effect of salt concentration on the electrolyte characteristics. This battery is found to deliver a capacity of 78.405 mA h g −1 at 25 ☌ and 49.083 mA h g −1 at 0 ☌ with a capacity retention of 71.83% after 200 cycles, demonstrating its potential for operations at low temperatures. Additionally, a full battery is assembled using the PPy cathode and PANI anode coupled with 19 m NH 4CH 3COO WiSE. Ammonia atom (NH3) reacts with hydrogen ion (H+) to produce ammonium ion (NH4+) by forming a coordinate covalent bond. When tested in a three-electrode cell containing 25 m NH 4CH 3COO electrolyte, PPy exhibits an impressive capacity of 125 mA h g −1 at a specific current of 1 A g −1 and retains 43.61 mA h g −1 at 25 A g −1. Predict the type of bonding in the following molecules : (i) Oxygen (ii) Calcium oxide (vii). For the first time, PPy is demonstrated as a high-capacity host material for both NH 4 + and K + storage, when cycled in water in salt electrolytes (WiSEs). Metals can only lose electrons to provide positive ions. 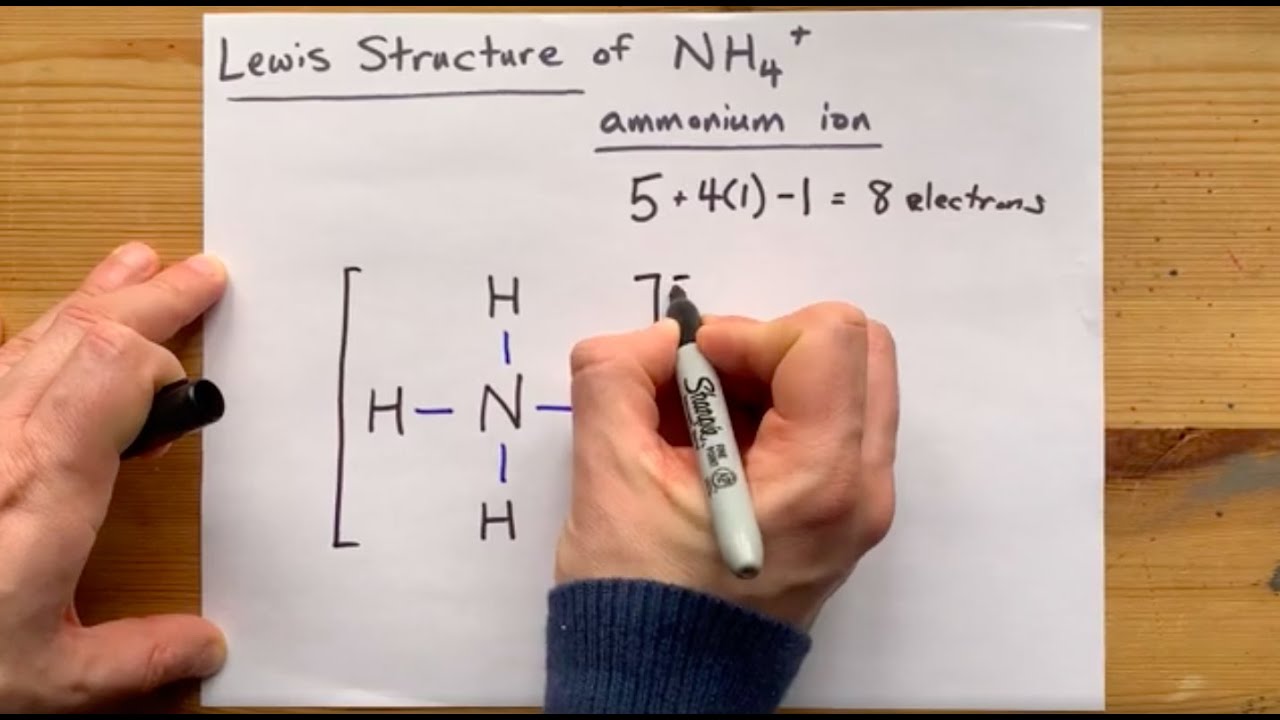
For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of Figure 22.4. Herein, we present an all-organic metal-free NH 4 + ion full battery that can operate at a low temperature of 0 ☌, by using polypyrrole (PPy) as the cathode, polyaniline (PANI) as the anode, and 19 m ammonium acetate aqueous solution as electrolyte. Ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. Thus, metal-free batteries present a unique opportunity as sustainable energy storage devices, though the relevant research is still in its infancy. Current commercial batteries are mainly metal based, with metal elements in charge carriers and/or electrode materials, which poses potential economic and environmental concerns due to the heavy use of nonrenewable metals.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
